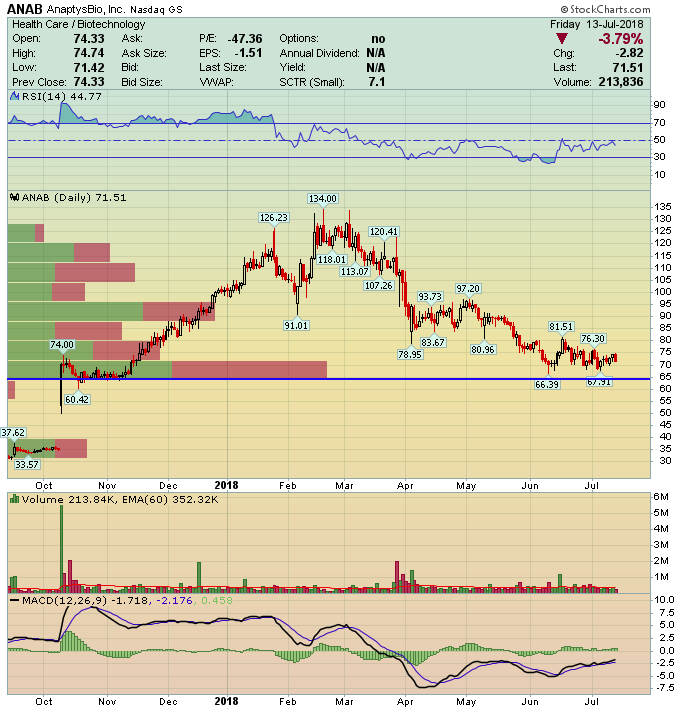
AnaptysBio (ANAB) – Bated Breath
AnaptysBio (ANAB) is a San Diego-based clinical stage biotechnology company developing antibody therapies for the treatment of inflammations and immune-oncology.
The company’s product pipeline includes wholly-owned ANB019 and ANB020, and a few other therapies that are being jointly studied with TESARO (TSRO) and Celgene (CELG).
At this time, will be focusing our discussion on ANB020 and its three intended indications.
Methodology
Contrary to other developments to date treating similar conditions, ANB020 targets interleukin-33 (IL-33) inhibitor to potentially suppress cytokine releases that lead to inflammation. The IL-33 key cytokine lies upstream of others, including IL-4, IL-5 and IL-13. By targeting the upstream IL-33, ANB020 affects all cytokines downstream which the company contends and has shown to be a better avenue than individually targeting the others. Regeneron (REGN) in partnership with Sanofi, is also in early trials of IL-33 based research, currently in Phase I and planning to initiate Phase II during 2018.
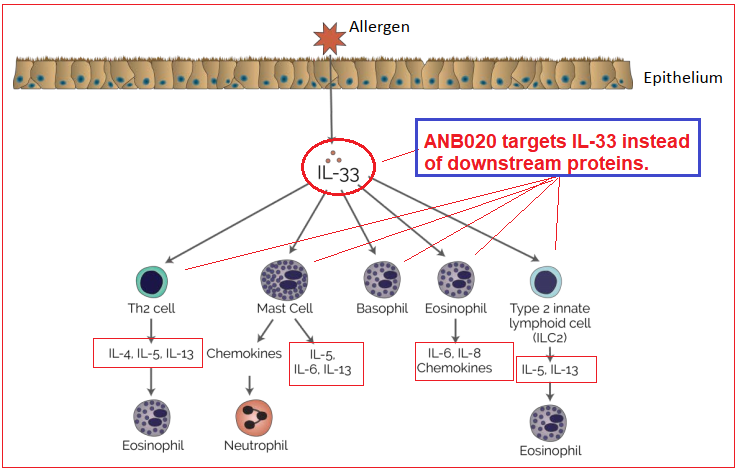
Cytokines are small proteins produced by a wide range of cells, including immune cells that are important in cell signaling. Interleukin are also proteins, playing an anti-inflammatory role in the immune system.
Options Activity
Recently a small flurry of bullish options activity caught our attention. Summarized are the transactions and dates:
July 12th –
- 150 in the money October 80 puts sold to open for $15.45 credit, approximately $231,000 put premium collected on wide bid/ask spread.
- 330 August 80/100 call spread bought for $4.10 debit, approx $143,500 bullish bet on 9x daily average call volume.
July 11th –
- 400 September 90 calls bought for $5.30, aggressive $212,000 bullish bet on wide bid/ask spread.
Upcoming Catalyst in Asthma
AnaptysBio is currently conducting a Phase IIa trial of ANB020 for Severe Adult Eosinophilic Asthma with topline data expected in 3Q2018. Twenty-four adult patients are taking part in this efficacy assessment study measuring improvement in Forced Expiratory Volume one second after administration of a single dose of ANB020 or placebo. This measures how much air a person can exhale during the test, and by calculating the patient’s total amount of expelled air, lung function can be evaluated to ascertain how well a specific treatment used to improve breathing is working.
Based on previous results demonstrated by ANB020 applications, expectations are for the
drug to show benefits vs. placebo during the trial. Jefferies analyst cites that his research suggests most biologics deliver 6%-9% improvement in lung function, and if this study can deliver a 7%-8% improvement, shares are likely to see $15-$20 of upside, and higher if results are better. On the downside, a miss on improvement could cause shares to stumble lower by $20-$25. Into this catalyst, he maintains a Buy rating and $145 price target.
JMP also remains a buyer on any weakness going into the top-line proof of concept release. The analyst maintains an Outperform rating and $180 price target on the shares, and has assigned about $61/share for the eosinophilic asthma indication alone.
RBC Capital’s analyst expects ANB020 to show a benefit over placebo in the trial, albeit with a caution that the data may be ‘challenging to interpret’ vs. competitors existing and emerging products.
Atopic Dermatitis
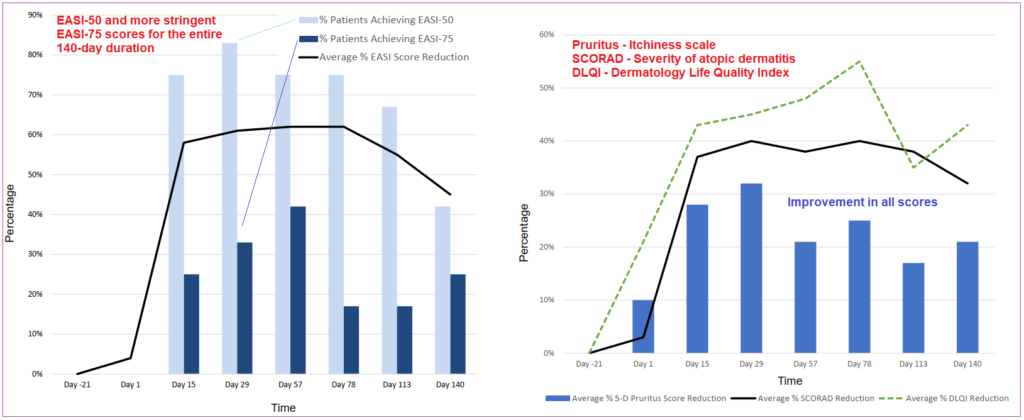
In early October 2017 shares soared 100% on ANB020 proof-of-concept in Atopic Dermatitis (eczema) Phase II topline data results that showed clear evidence of lasting disease remission. Furthermore, none of the twelve patients in the study reported any major safety concerns.
All subjects were administered a single, 300mg intravenous dose of the drug. Relative to their enrollment baseline, 83% of the patients achieved EASI-50 (Eczema Area and Severity Index) score at day 29, and 75% achieved that score on day 57, this is the standard tool used by medical field to measure the severity of eczema. The trial also showed ANB020 impeding Th-2 immune cell-driven inflammatory responses averaging 30% across the study, reaching as high as 60% on patients that achieved EASI-50 at day 20 checkpoint.
Although the study consisted of just twelve patients, all of them showed improvement within the predetermined timeframes, a strong response which cannot be disregarded. During the first half of 2018, AnabtysBio provided further updates on the study and furthermore initiated a Phase IIb randomized, double-blinded, placebo-controlled study in 300 adult patients with moderate-to-severe atopic dermatitis. The goal of this trial is to evaluate multi-dose subcutaneous administrations of ANB020; results are expected sometime in 2019.
Peanut Allergy
In late March, AnaptysBio released early data from its Phase IIa proof of concept trial on 20 adults with peanut allergies. The results showed that 46% of patients receiving a single dose of ANB020 were subsequently able to tolerate a cumulative 500mg dose of peanut protein two weeks later. In contrast, none of the patients receiving placebo were able to reach that level of tolerance.
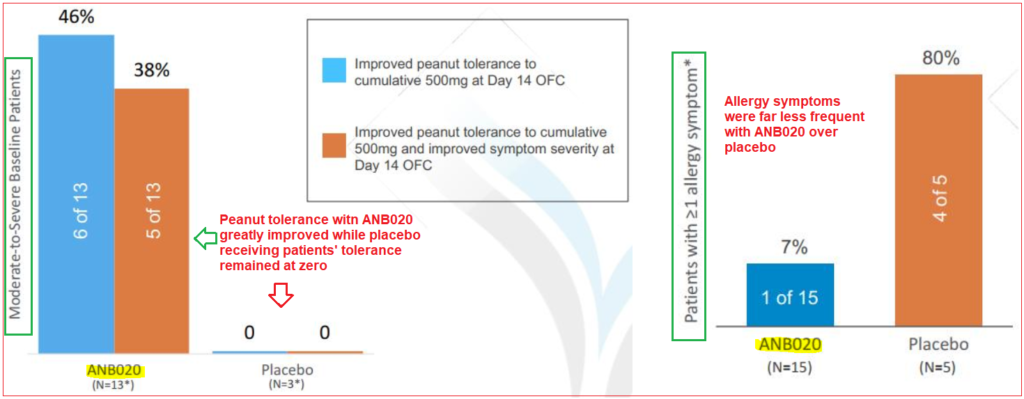
While the trial results were positive, an issue that many investors and analysts had post-presentation was the company’s decision to exclude four participants from the study’s analysis due to mild allergy symptoms; this led to concerns that the treatment’s true potential was being artificially elevated and is reflected in the direction of the share price since the release. Furthermore, certain peanut allergy specialists who reviewed the data questioned the company’s classification of patient symptomology (defined as “mild”, “moderate” and “severe”) does not exist in current guidelines, applying only to oral food challenge (OFC) situations. RBC Capital sees this subgroup analysis as questionable, remarking that investors’ skepticism and credibility concerns towards management have increased. After the data was presented, the analyst reduced his price target from $144 to $86.
However not all firms were of the same mind, Stifel cited the trial’s positive results, lack of discontinuations and no serious adverse events leading to expectations of longer-duration trials with multi-dosing structures that will lead to increased peanut tolerance. The analyst raised his target price to $152 from $139. Credit Suisse also remained bullish, stating that the results had increased their confidence. The analyst did mention though that more confidence would be gained from a larger data set as the small number of this group were “too small to make a definitive conclusion”, but would be supportive of studies on guidelines established or based on PRACTALL.
Closing Thoughts
AnaptysBio’s shares have drifted lower since late March on its peanut allergy results. To recap, it wasn’t a lack of viable data, rather the manner in which management decided to classify severity levels and also the dismissal of four datasets that did not endear to investors. It is possible that the projected release of eosinophilic asthma trial results will set the company back on track, not only showcasing ANB020 efficacy and safety, but also rebuilding investor confidence in the company. This could potentially be the stock’s turning point, albeit with some speculative risk.