Axsome Therapeutics (AXSM) – Two Readouts in 1Q
Axsome Therapeutics (AXSM) is a clinical-stage biopharmaceutical company developing novel therapies for the management of central nervous system (CNS) disorders, including pain, Alzheimer’s disease and depression. Founded in 2012, the company is headquartered in New York City.
Recent Developments
On January 7th, shares jumped higher by as much as 230% after the company announced AXS-05 Phase II trial in major depressive disorder (MDD) having met its primary endpoint.
Results of the randomized, double-blind, active controlled multi-center US trial on moderate-to-severe MDD patients who were treated twice a day for six weeks demonstrated a “highly statistically significant” reduction on Montgomery–Åsberg Depression Rating Scale (MADRS), a scientific questionnaire designed in 1979 by British and Swedish researchers and used to measure the severity of depressive episodes in patients with mood disorders.
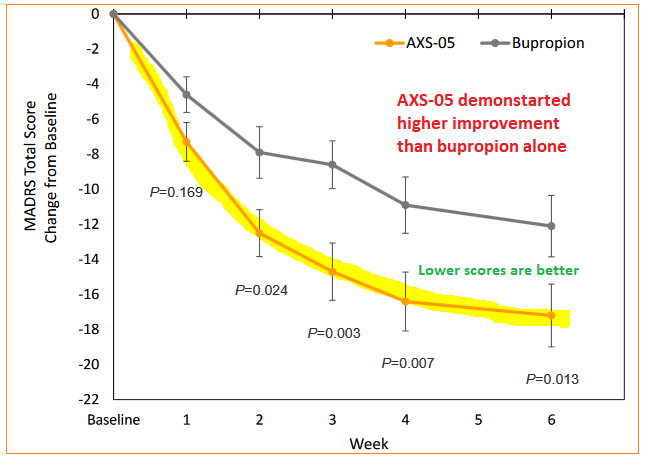
At Week 6, AXS-05 had a 17.2 point reduction versus 12.1 points for bupropion – the active comparator, a medication primarily prescribed as an antidepressant or smoking cessation aid, either on its own or in conjunction with other medications. Also on Week 6 time frame, AXS-05 had a 47% remission rate while bupropion-only patients showed a 16% rate.
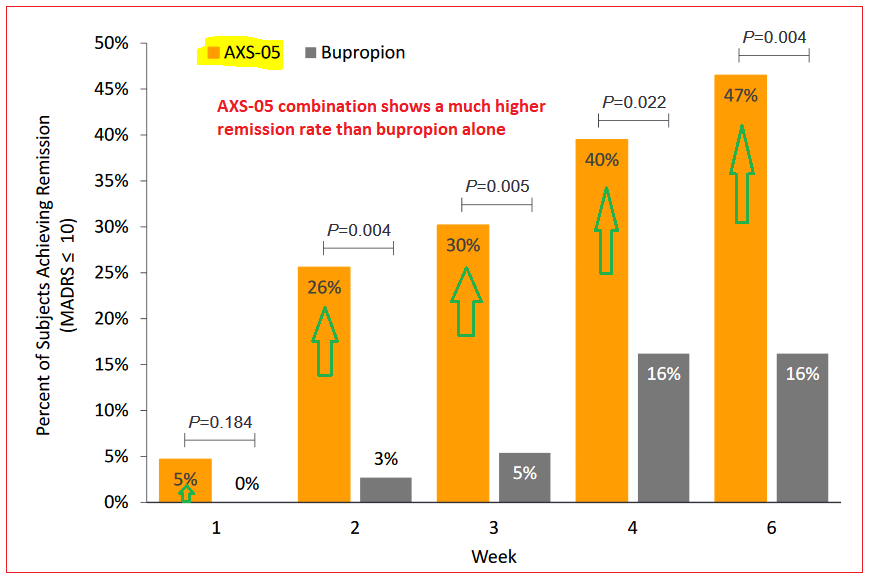
The complete results of ASCEND study will be presented at upcoming scientific symposiums during 2019. Meanwhile based on the positive outcome to date, management plans to meet with the FDA with the goal of expanding the treatment to a broader application.
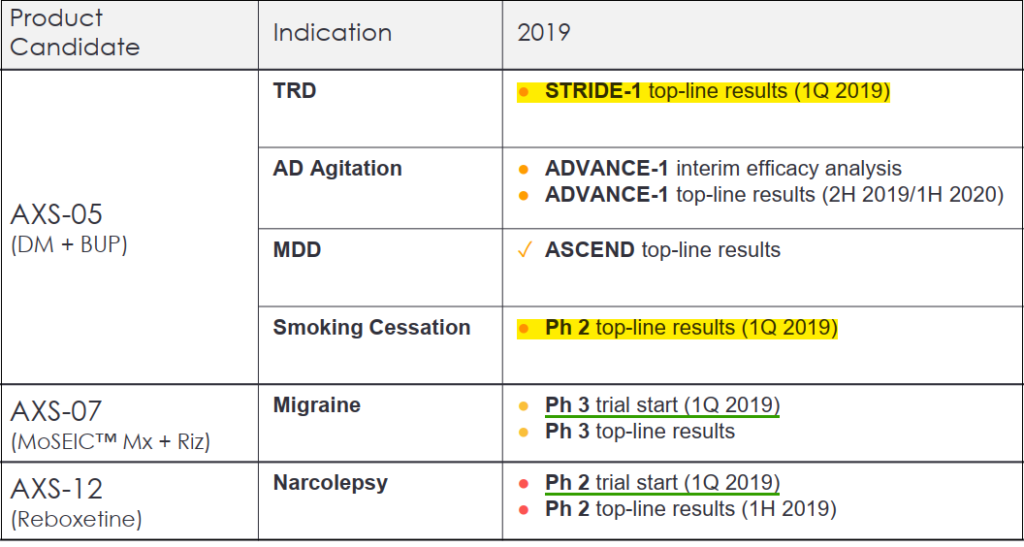
Pipeline Catalysts
Although Axsome has multiple therapies in development, we’ll focus on the two that have the closest catalyst dates. Both of these trials are for AXS-05 which is a combination of bupropion and dextromethorphan (DM) formulated to treat different indications. Results for both these trials are expected in 1Q2019.
– AXS-05 STRIDE 1 Phase III study for treatment resistant depression (TRD), a randomized, double-blind active control trial (similar to MDD study mentioned earlier) targeting patients aged 18-65 who previously had inadequate response to one or two other antidepressant treatments.
Based on the results of interim futility analysis of the first 100 patients (of 250), an Independent Data Monitoring Committee (IDMC) concluded AXS-05 appeared safe and well-tolerated and recommended that the study continue to full enrollment. Back in early 2017, the FDA already had granted TRD a fast-track designation due to the serious nature of the condition and its unmet medical needs.
As the first interim results demonstrated safety and tolerance in patients, the next dataset will be looking for efficacy using MADRS. If the overall results are positive, the study will continue with a second Phase III trial. Based on ASCEND’s outcome in January, analysts believe STRIDE-1 will be successful.
– AXS-05 Phase II study for smoking cessation in collaboration with Duke University. The trial began in April 2018 involving 60 smokers willingly attempting to quit the habit. The primary outcome measure of the double-blind, randomized, controlled study is to achieve a change in smoking intensity.
In preclinical trials, DM demonstrated the desired positive effects in rats. In humans, AXS-05 DM-bupropion combination caused a significant increase in DM plasma concentrations which is the intent as studies have determined that that both DM and its metabolite dextrorphan block nicotinic receptor function in a noncompetitive but reversible manner, suggesting that both drugs block the receptor channel helping those that are addicted to nicotine refrain from smoking. Side note, DM has been used as a cough suppressant for nearly six decades (hence the DM wording on cough medicine), it also has been reported to be useful in helping with heroin withdrawal.
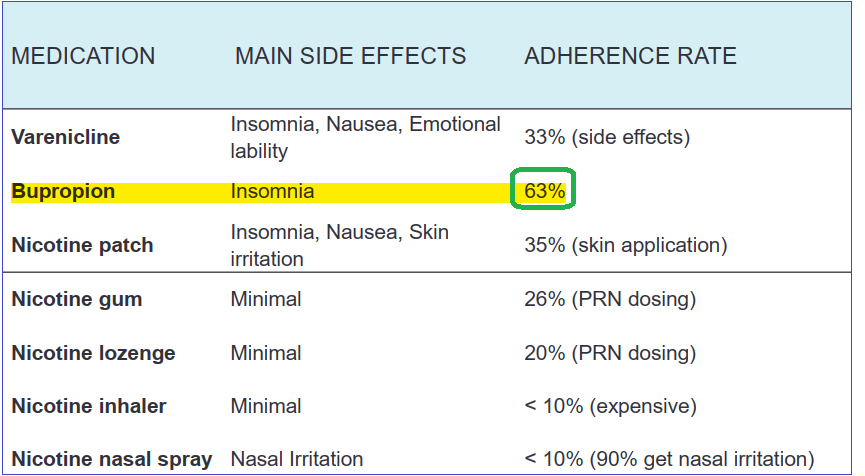
The smoking cessation market is very large and continues to increase. In the US alone, there are around 40 million smokers and nearly 30% of them actively attempt o quit using medications: overall, nearly 70% of smokers have indicated their desire to quit. Although there are a few approved cessation aids on the market, some have serious side-effects, such as varinecline, marketed by Pfizer (PFE) under Chantix and Champix while others have shown varying rates of program adhesion. Of all therapies, bupropion has seen the highest adhesion rate by patients.
Analyst Ratings
Axesome doesn’t have very wide coverage, the most current analyst ratings are as follows:
- Cantor Fitzgerald initiated at Overweight with a $16 target in November 2018
- HC Wainwright rates at Buy with a $15 target, increased from $10
- BTIG rates at Buy with a $20 target, increased from $16
- Ladenburg Thalmann rates at Buy with a $45 target, increased from $34
Looking Further
Along with the two indications mentioned above, AXS-05 is being studied for Agitation in Alzheimer’s Disease (AD) where topline results are expected in late-2019 or early 2020. Two other therapies are also in the pipeline, AXS-07 for migraine and AXS-12 for narcolepsy – both these studies will begin in 1Q2019 with the narcolepsy trial targeting 1H2019 (likely 2Q) for results.