Behind The Numbers – Natera (NTRA)
Natera is a rapidly-growing diagnostics company with proprietary bioinformatics and molecular technology for analyzing DNA. Since 2009, Natera has launched seven molecular diagnostic tests, many of which are available through major health plans accounting for more than 140 million covered lives in the United States.

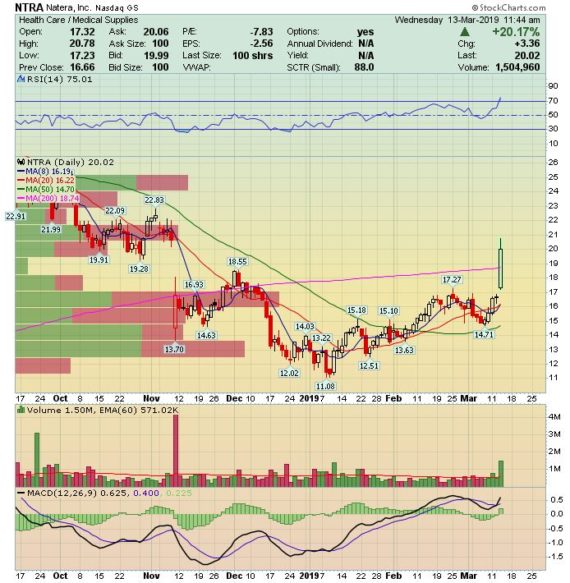
The stock is surging today, up over 20%, after reporting Q4 earnings after the bell yesterday:
-EPS of ($0.51) vs ($0.52) estimate – Beat
-Revenue of $67M vs $63.97M estimate – Beat
-Total Revenue increased 23% Y/Y
-Tests Processed increased 30% Y/Y
“2018 was a very successful year for Natera,” said Steve Chapman, Natera’s Chief Executive Officer. “We grew revenues substantially over 2017, doubled our volume growth rate year over year, and delivered compelling data in both oncology and in kidney transplant rejection. We see 2019 as a key inflection point for Natera across reproductive health, oncology, and transplant.”
A Promising Pipeline
Signatera – On the conference call, the company announced a $50M partnership with BGI Genomics to leverage their extensive region operations to bring Signatera to the Chinese market. CEO Steve Chapman said, “We believe China represents a very large clinical testing opportunity, and we also expect this partnership to support the adoption of Signatera into a pharma-sponsored clinical trial in China. This may give us an advantage with global pharma customers running trials in China since Chinese law prohibits the export of patient blood samples overseas. BGI already enjoys a significant leadership position in genetic testing in China. For example, last year, they performed more than 1 million cell-free DNA test. This partnership with BGI strengthens our oncology franchise, expands our reach in reproductive health in select markets and may reduce COGS in our lab.”
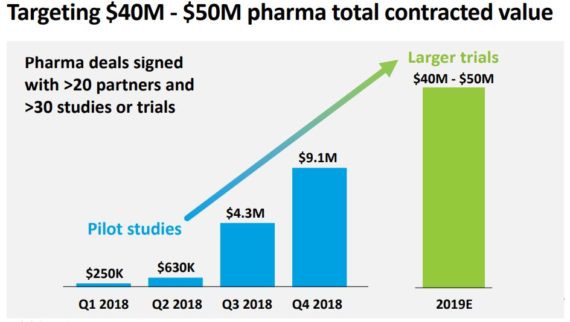
In addition to the BGI deal, the company also highlighted that their Signatera commercial efforts continue to accelerate. They started 2018 with just a few hundred thousand dollars in signed Signatera pharma services contracts and continue to evolve by signing several prospective clinical trials with leading pharmaceutical companies. “The bars on the page represent the total contracted value, which should be the amount of revenue we’d expect to recognize over time from signed deals based on the pricing and the test volume specified at each contract. We generally expect most signed deals to translate to recognized revenue over roughly 12 to 36 months depending on how long it takes for prospective clinical trials to enroll patients, among other factors.”
Natera believes 2019 can be an inflection year for Signatera. They believe they can secure signed pharma contracts with accumulative value of $40M – $50M by the end of 2019. This would be a major step forward for the business and sets them up for significant revenue growth.
Transplant Testing – On the conference call, Natera discussed its critical milestones that will help them reach a “tipping point” for this business. This morning, JPMorgan analyst Tycho Peterson summarized it best:
“Turning to the kidney transplant rejection test, NTRA reiterated timelines for commercializing the test and securing Medicare reimbursement by YE19, while also highlighting studies validating the accuracy of the test, including the recent analytical validation study published in Transplantation indicating that NTRA’s assay has greater “area under the curve” (0.87 vs 0.74), performs better in T-cell mediated rejection cases (about 1/3 of all cases) and is the only test that works in sub-acute clinical rejection (~20-25% of cases). Following LCD submission, management expects first the draft LCD release, followed by the final LCD, after which NTRA plans to commercialize the test, complementing its own direct sales channel through partnering with One Lambda, a division of TMO, to leverage their strong commercial infrastructure including the largest U.S. transplant centers.”