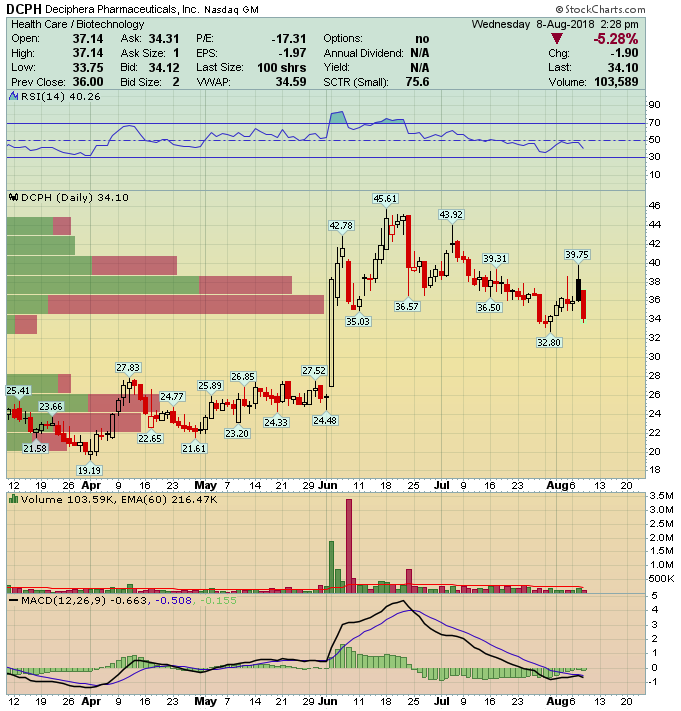
Deciphera Pharmaceuticals (DCPH) – Trio of Cancer Trials
Massachusetts-based Deciphera Pharmaceuticals (DCPH) is a clinical-stage biopharmaceutical company developing drugs to improve the lives of cancer patients.
Its current pipeline is comprised of three drugs DCC-2618, DCC-3014 and Rebastinib. All three are orally-administered treatments that have shown promising results to date targeting different cancers.
DCC-2618
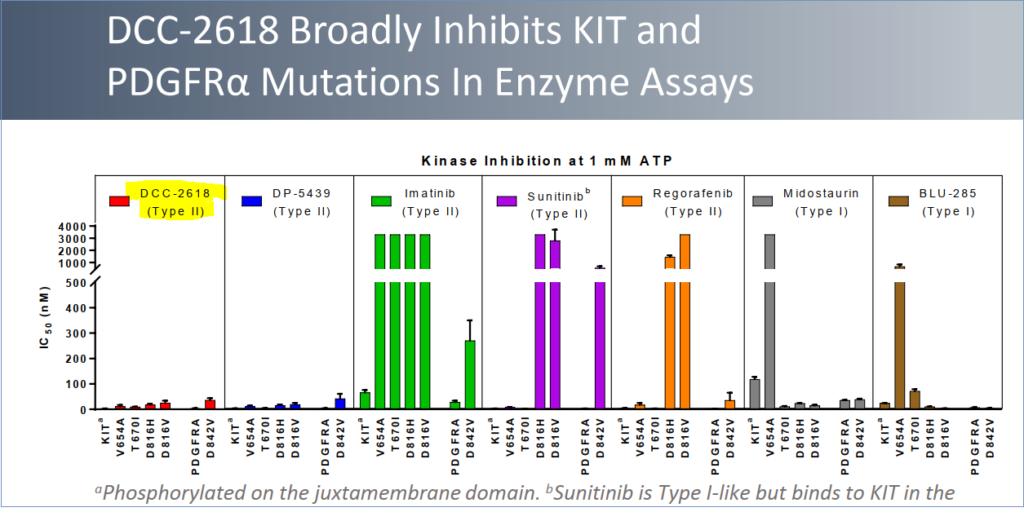
Being developed for the treatment of gastrointestinal stromal tumors (GIST), DCC-2618 is the lead candidate of Deciphera’s clinical trials. It is a kinase switch control inhibitor that is also being tried on advanced systemic mastocytosis (ASM) and other solid-tumor cancers as well, specifically those driven by KIT or PDGFRα kinases. A kinase is an enzyme that helps transfer phosphates within the body’s cells, and various cancers can result when this enzyme is mutated.
During this year’s American Society of Clinical Oncology (ASCO) meeting in early June, updated interim Phase I results were released that showed very positive data in second, third and fourth-line heavily pre-treated GIST patients, and on which the company announced a further, Phase III trial, INTRIGUE, in second-line GIST patients during 2018. Compared to existing, approved treatments, DCC-2614 showed superior efficacy in 2nd and 3rd line patient treatment, and also in 4th line where there aren’t any current approved therapies.
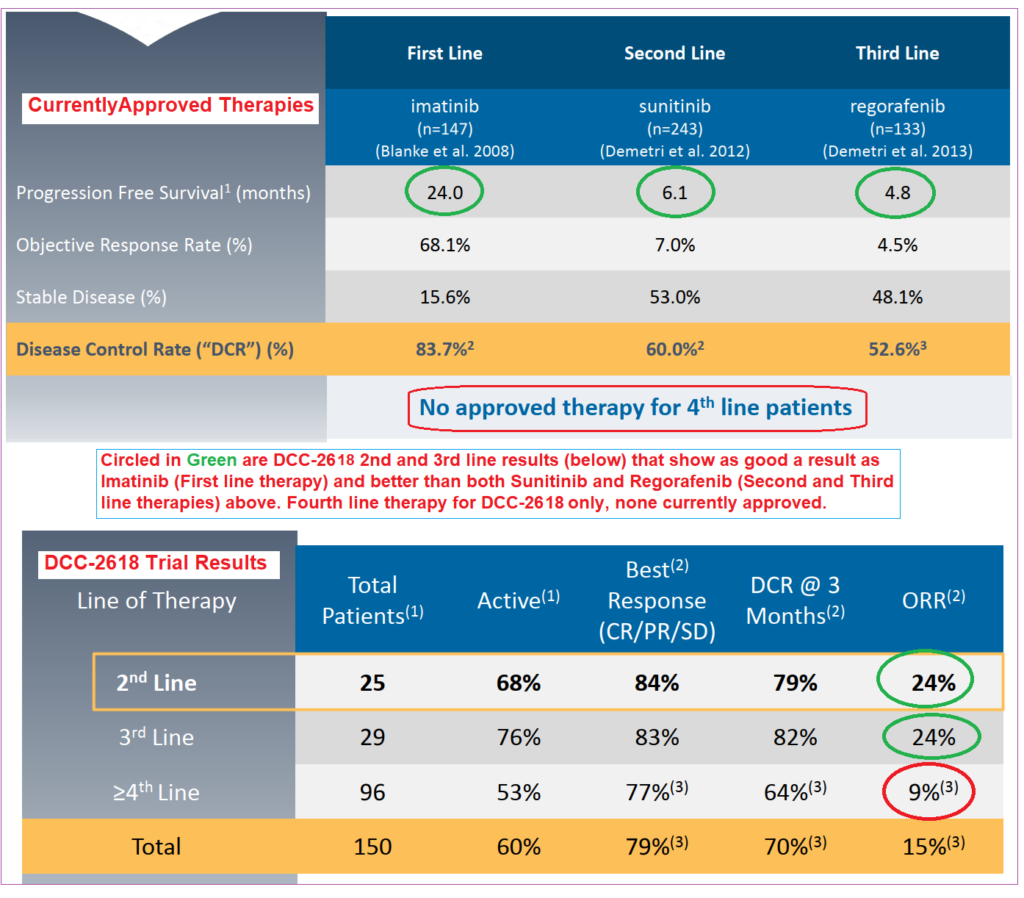
Combined with previously-released tolerability data in early 2018, overall results demonstrated the potential of DCC-2618 to be an effective and well-tolerated treatment in GIST. 78% of patients (57 of 73) receiving daily doses of 100mg or more showed a KIT MAF (gene mutation) reduction registering higher than 50%.
DCC-3014
A solid tumor treatment, DCC-3014 is an orally-administered highly selective therapy designed to bind to the unique switch socket in CSF1R which controls the function of macrophages, a type of white blood cell that helps the immune system by engulfing and digesting foreign substances, cell debris, microbes and cancer cells. CSF1R, for the non-medical specialists among us, stands for colony stimulating factor 1 receptor. It is a receptor for cytokine (a small protein) that is important for cell signaling, and especially significant in the immune system
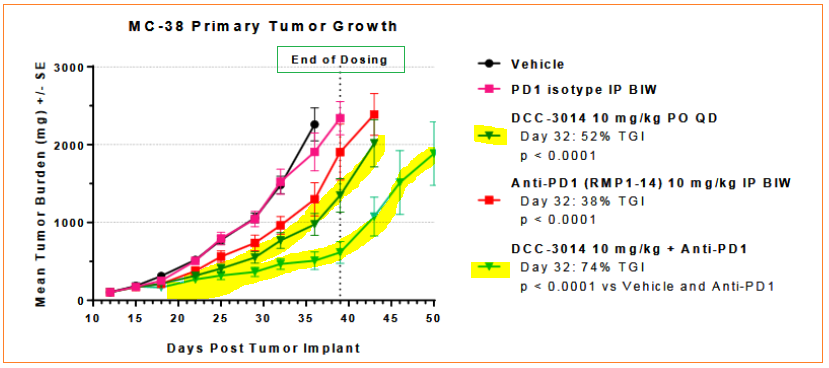
In a preclinical model, DCC-3014 was evaluated alone and also combined with an anti-PD1 antibody, part of an emerging class of cancer treatments. The effectiveness of DCC-3014 together with the anti-PD1 antibody demonstrated greater results than of a vehicle control, control antibody or the stand-alone ant-PD1 antibody treatment itself.
As a result of the preclinical trial’s readouts, DCC-3014 was moved into a Phase I clinical study in patients with advanced malignancies where tumor-associated macrophages are confirmed or believed to be enabling further growth and spread of the cancer.
Preclinical trial of DCC-3014 was performed on colorectal cancer cell model, the results of which showed additive beneficial effects with an anti-PD1 antibody in blocking tumor growth and promoting immune system activity:
- Anti-PD1 antibody on its own showed 38% tumor growth inhibition
- DCC-3014 by itself showed 51% tumor growth inhibition
- DCC-3014 + anti-PD1 antibody showed 74% tumor growth inhibition
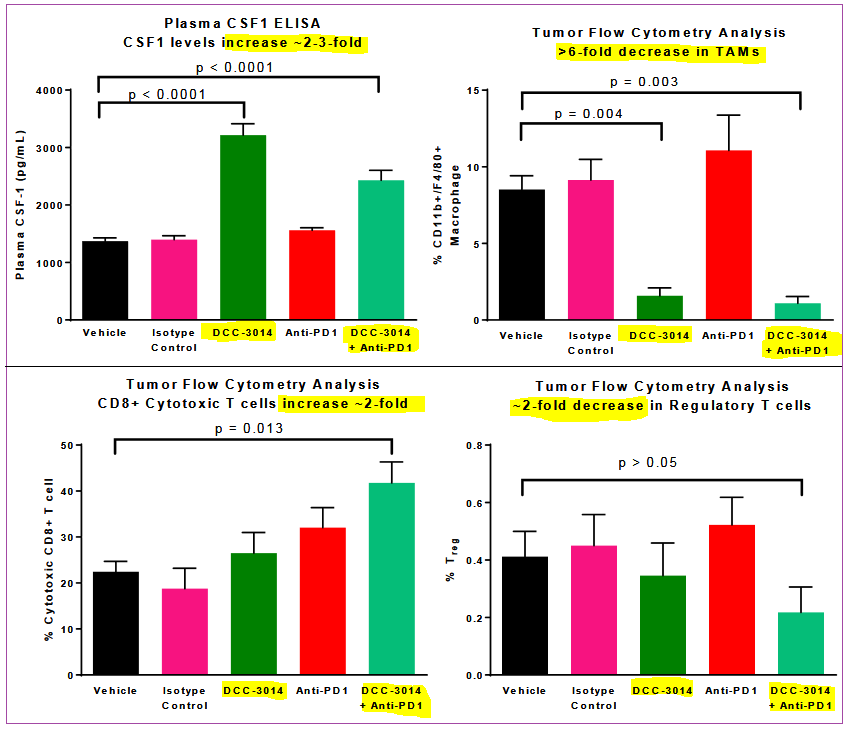
Tumor-associated macrophages were greatly reduced in the primary growth, and cytotoxic CD8+T cells (cancer killing cells) increased about 2-fold in the combination treatment group.
Deciphera is expected to provide a Phase I Dose Escalation data update in 2H2018.
Rebastinib
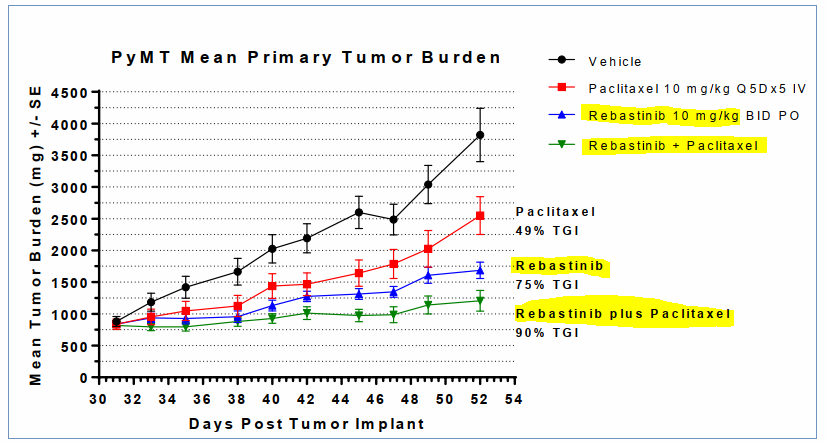
A treatment being developed for breast cancer, Rebastinib is currently undergoing Phase I testing on solid tumors in combination with chemotherapy.
Early results have shown tumor growth inhibition as a stand-alone therapy with better response than Paclitaxel chemotherapy drug. When combined together, the tumor growth inhibition was even higher.
Analyst Ratings
Five of the six analyst firms covering Deciphera are bullish, the sixth is neutral.
- Canaccord initiated at Buy on August 7th, 2018 with a $55 price target
- SunTrust rates it a Buy
- JMP Securities rates it Outperform with a $65 price target
- B. Riley downgraded from Buy to Neutral in June with a $43 price target
- Piper Jaffray rates it Overweight with a 50 price target
- JPMorgan rates it Overweight with a $33 price target
Closing Thoughts
Deciphera is yet to be a well-known name in the cancer research field, on average it trades around 280,000 shares per day and does not have options at this time. Its three development products have shown good promise in their early stages of testing without any reported side-effects.