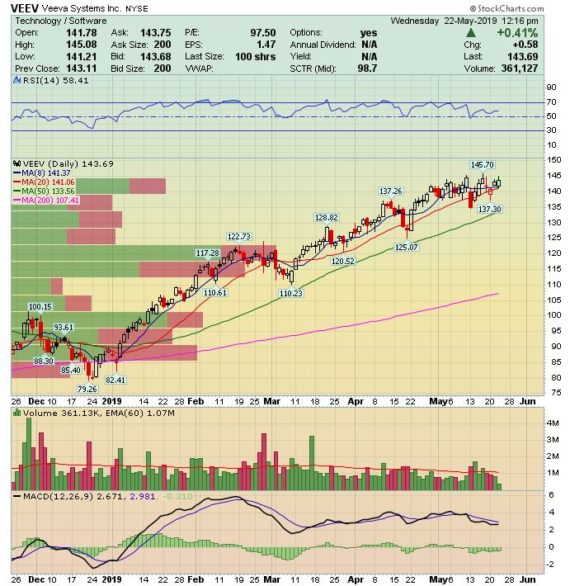
Veeva Systems (VEEV) – Upgrading The Vault
This morning, Stifel analyst Tom Roderick raised his price target on VEEV to $150 from $130 with the synopsis being that the company’s Clinical Data Management and Clinical Operations suites hold the highest potential for long-term growth for the company. Despite the presence of numerous competitors, Stifel says their industry checks suggest that Veeva has made substantial progress in both its product and go-to-market efforts with these solutions since their launch just a few years ago.

Stifel would expand on their commentary by saying that in a recent report, competitor IQVIA highlighted a record number of drug approvals in 2018 (59 novel treatments) and a success rate of ~11% for drugs entering the clinical development stage. Stifel interprets these figures as a tailwind for Veeva’s business, as the company’s product suite is one of the only in the market that addresses the needs of Life Sciences organizations from infancy (clinical trials) to maturity (promotional cycle).
Industry Checks
CDMS – Despite CDMS being a newer category for the company, Stifel’s checks suggest that Veeva has finally begun to achieve solid footing in the market and stands up well competitively to incumbents like Medidata Solutions (MDSO) and Oracle (ORCL) Phase Forward. This feedback is notably different from what they were hearing this time last year, as some Tier 1 pharmaceutical customers they spoke with indicated the platform did not scale appropriately at the time, and that functionality paled in comparison to Medidata and Phase Forward. That feedback has reversed course meaningfully in their most recent conversations with industry experts who are now seeing Veeva in some Phase 2 trial deployments, with functionality that has come a long way.
CTMS – Stifel is also looking for Veeva to sustain its momentum in the Clinical Operations space through the attachment of CTMS (Clinical Trial Management System) and Study Startup. In the case of CTMS, Veeva secured its first Top 20 win last F3Q and doubled its customer base last year to 34. Their checks have consistently and uniformly indicated that the Trial Master File (TMF) roadmap is one that has very natural overlap to CTMS. As one contact said, “All systems have TMF reference models for trial documents.” In other words, all CTMS patient data (Routine Monitoring Visits, Trip Reports, Financial Disclosures, and essentially all other relevant details of the patient visit) can be used for site-level documents. Like CDMS, CTMS also seems to be making solid strides in application improvement. With three upgrades a year, the opportunity to bolster functionality is ongoing. For example, one such customer request that they hear is in beta at Veeva is Site Payments, and they continue to pick up on new integrations between CTMS and other core health systems.
Finally, Stifel adds that Veeva’s efforts outside of Life Sciences have already yielded strong returns, with the company nearly doubling its customer base last year. Recently, the company unveiled Veeva Claims, an end-to-end product and marketing claims management module for organizations outside of its core Life Sciences base. The solution provides workflows for the entirety of the claims process and unifies organization’s legal, regulatory, research and development, and marketing teams on a common dashboard. With Claims, Veeva aims to reduce friction between teams during the claims process and provide a full view of individual claims through a detailed audit trail. “We anticipate additional details on the progress of QualityOne and newer solutions like Veeva Claims on the company’s conference call.”
Q1 Earnings will be taking place on May 29th After the Close
JaguarScan
According to JaguarScan, here are two notable trades that remain in open interest:

-On 3/21, there was a buyer of 100 June 130 Calls for $7.50, a $75,000 bullish bet.
-On 5/10, there was a buyer of 100 January (2020) 160 Calls for $13.20, a $132,000 bullish bet.