Spero Therapeutics (SPRO) – 2H2019 Readouts
Spero Therapeutics (SPRO) is a multi-asset, clinical-stage biopharmaceutical company focused primarily on new antibiotic formulations and also on improving existing antibiotic effectiveness via their Potentiator platform. Founded in 2013, their IPO was in November of 2017. Spero’s market cap is around $185M.
Of four clinical-stage products in their pipeline, three are expected to provide trial updates in 2H2019:
- SPR994 Phase III interim data
- SPR206 Phase I data
- SPR720 Phase I data
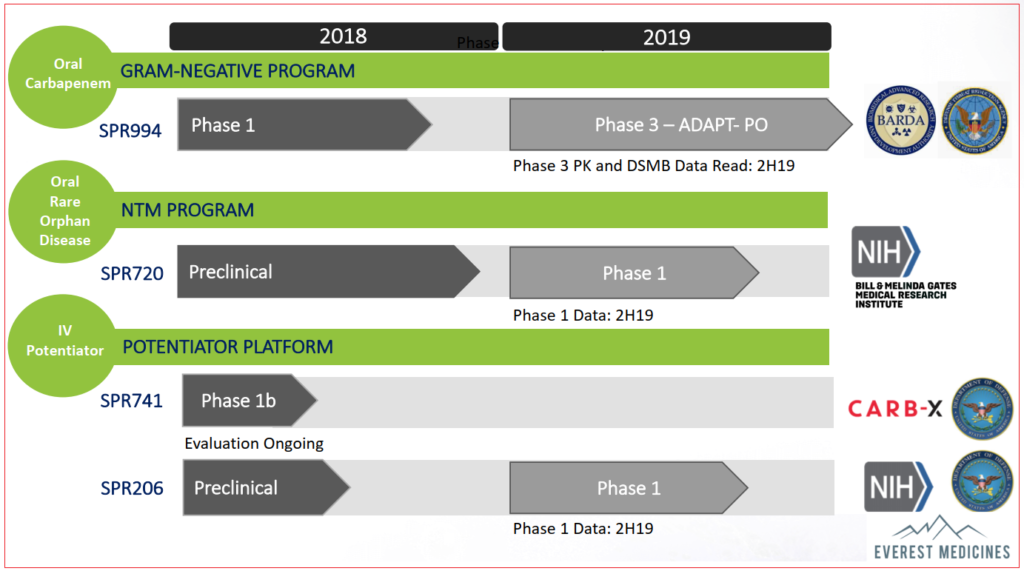
SPR994
Currently undergoing Phase III randomized, double-blind evaluation, SPR994 is an oral therapy of tebipenem for complicated urinary tract infections (cUTI). Tebipenem isn’t an altogether untested product itself, it has been marketed in Japan since 2009 under the brand Orapenem for common pediatric infections such as pneumonia and sinusitis: however this is an intravenous delivery whereas current trials are for oral administration.
SPR994 is
