Global Blood Therapeutics (GBT) – Sickle Cell Fighter
Global Blood Therapeutics (GBT) is a clinical-stage bio-pharmaceutical company that is developing a novel drug for Sickle Cell Disease treatment. Based out of San Francisco, the company has a market cap of $1.7 billion.
Sickle Cell Disease (SCD) does not have any preventative treatments. Blood transfusions and Hydroxyurea (a drug that inhibits the production of DNA) are the most commonly used therapies yielding modest reductions to the re-occurrence of painful effects common to the disease. However the use of these treatments is often restrained, or not used in any optimal fashion as it contributes to anemia and reduced white blood cells which are important in fighting off infections. As well, a significant number of patients are unable to tolerate hydroxyurea.
Global Blood Therapeutics’ GBT440 (voxelotor) is a pill-form small molecule hemoglobin modifier – it forms a reversible chemical bond with the hemoglobin’s alpha chain, causing enzymatic change that attracts more oxygen. The company’s one and only drug in development, it has been successful in its trials as of date. Phase III HOPE trial was begun in January of 2017 as a 2-part, randomized, double-blind and placebo-controlled multinational process which aims to enroll upwards of 400 patients (150in part A, ~250 in part B). Estimated completion timeline is June 2019 with production planned in 2020/2021.
On November 13th, the Independent Data and Safety Monitoring Board (DSMB) reviewed and confirmed the safety of the data. This opens up enrollment for younger patients, aside from being an important milestone for the HOPE study.
Clinical Data
Test results have been very encouraging to date. Data has shown increases in patients’ hemoglobin oxygen bonding levels in whole blood (hemoglobin is the iron-containing protein in the blood that transports oxygen to the tissues).
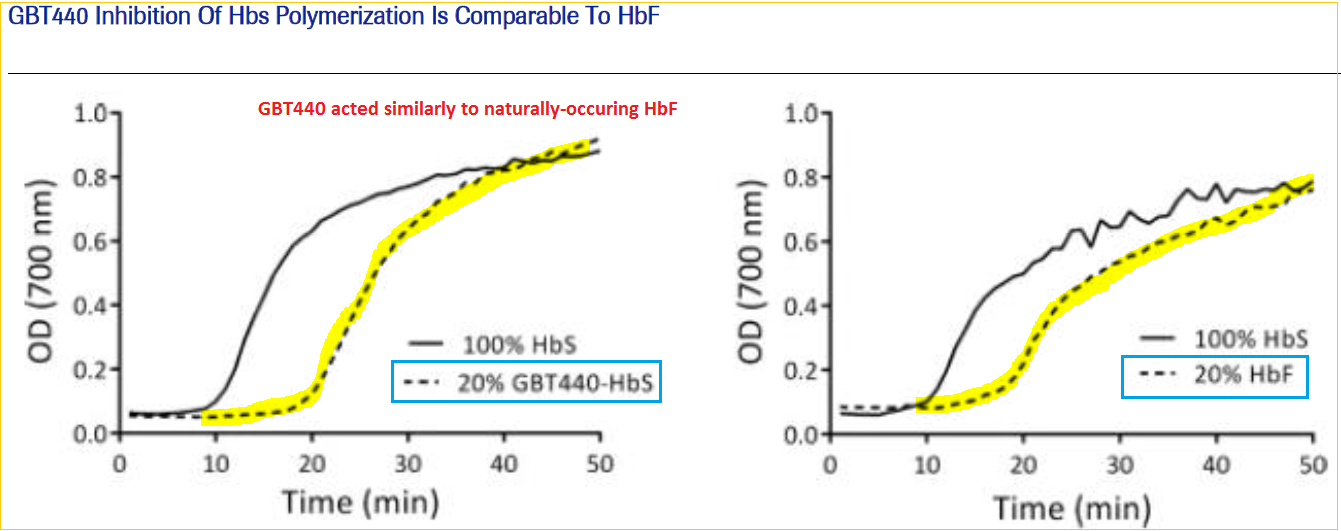
In-vitro results demonstrated that GBT440 acts in a similar fashion to naturally-occurring HbF (fetal hemoglobin), an inhibitor of deoxygenated HbS (abnormal type S hemoglobin) polymerization. Existing treatments target the downstream consequences of the diseases primary event, that is the unwanted polymerization (forming of chains) of hemoglobin. GBT’s novel approach treats the patients’ blood-oxygen levels at the core of the condition instead.
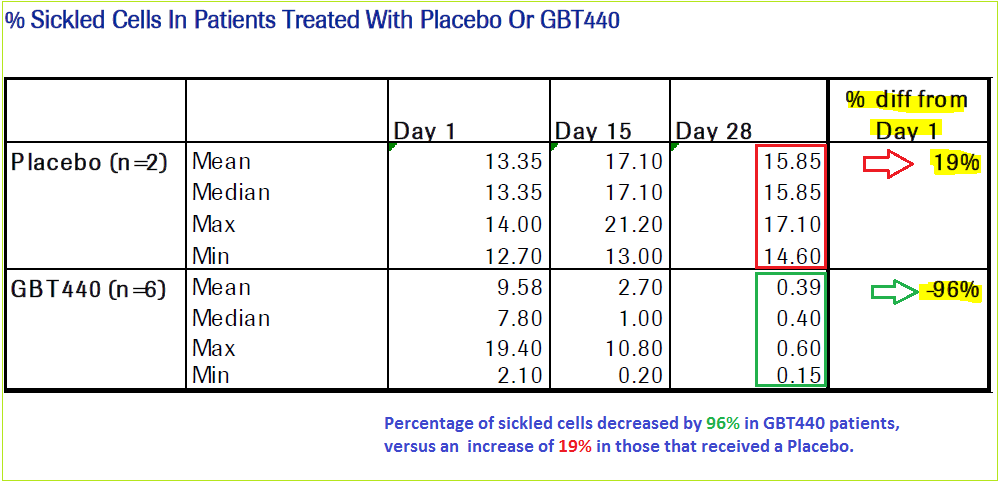
From its preliminary data collection, GBT440 once-daily 700mg treatment for 28 days on six patients showed a decrease in percentage of the sickled cells by 96%. Since the disease’s remaining characteristics are all downstream events, the potential of amelioration of all symptoms and resulting effects of SCD can be targeted.
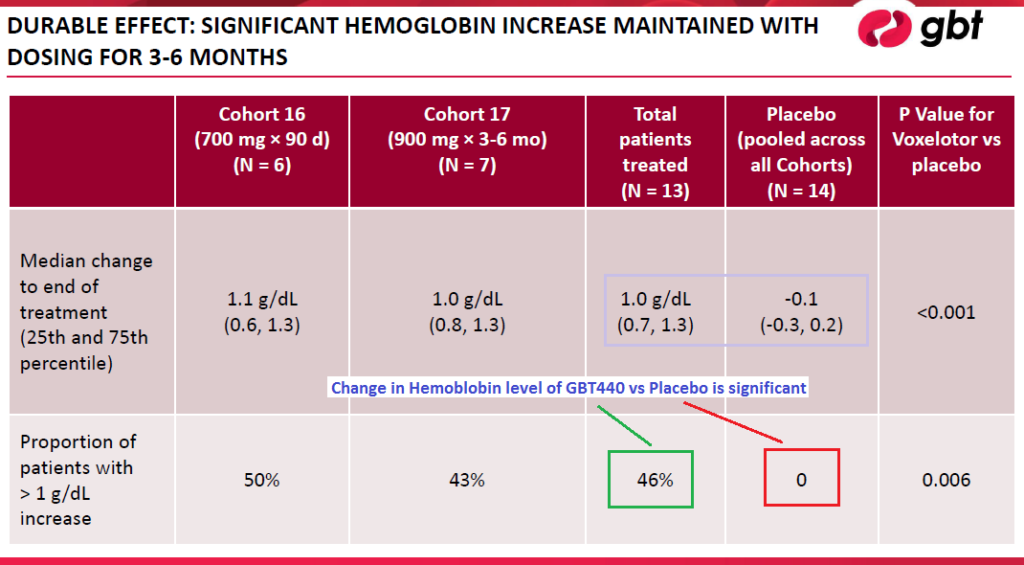
Results of the first 6-month GBT440 treatment period in 2016 showed increased levels of hemoglobin counts. Another positive finding was the decrease of unconjugated bilirubin – sufferers of SCD have higher levels of it as their red blood cells get broken down ~6x faster than normal red blood cells. Tests with a 900mg dosage level resulted in 42.9% decline versus the Placebo’s 2% increase. Additionally, to date there have not been any adverse effects of the drug, aside from low-grade rash reported on one patient and grade 2 diarrhea in another, neither of which reoccurred subsequently.
Overall, GBT440 was well tolerated up to 6 months of dosing. Adverse effects were in-line with Placebo patients complaints.
Parallel Trials
HOPE-KIDS 1 open-label single/multiple dose study evaluates the safety, tolerability and the effects of GBT440 on adolescents aged 12-17. The two-part study already has shown that a single 600mg dose was well-tolerated, with similar observations seen in adults. At this time, evaluations are being conducted in 900mg – 1,500mg dosages which, based on previous indications of lower dosages, should produce satisfactory results, a sentiment conveyed by Cowen’s analyst in a detailed research note from late September.
Another study is enrolling younger patients between the ages of 6 and 11. This trial should produce data in 2018 after proper dosing is fine-tuned, and a 6-month period of testing completed. Management noted that younger patients could potentially show even better results than older ones as their lower timeframe of exposure to the destructive effects of the disease is less pronounced.
Analyst Coverage
Recent analyst ratings and prices from September onward:
– Wedbush rates shares at Outperform with price target of $73
— Morgan Stanley rates shares at Overweight with price target of $56
– Needham & Company rates shares at Buy with price target of $51
– J P Morgan rates shares at Buy
– SunTrust Robinson rates shares at Buy with price target of $67
– Cowen rates shares at Buy with price target of $83
– Oppenheimer rates shares at Buy with price target of $53
2017 Timeline
- January 18th – First patient enrolled in HOPE Phase III study
- February 16th – Initiated Phase I trial in GST440 for physiological evaluations
- June 23rd – Announced new data to support ongoing clinical developments for GBT440 in adolescents
- July 10th – Expanded ongoing HOPE-KIDS 1 Phase IIa study in pediatrics
- September 5th – Received Rare Disease designation by the FDA for GBT440
- October 11th – Presented new data showing positive effects of GBT440
- October 28th – Presented case study at SCD Association of America Convention in Atlanta
- November 13th – Announced successful completion of DSMB’s safety review for HOPE Phase III study

Future Catalysts
GBT will be webcasting a presentation to be held on December 11th at the American Society of Hematology (ASH) 2017 Annual Meeting to be held in Atlanta, GA, from December 9th to the 12th. The company presentation is on December 11th at 20:15 Eastern time. It is expected that progress from GBT440 HOPE-KIDS 1 trials will be presented at the conference, this study is aimed at children aged 6 to 11.
HOPE Part A readout is expected in 1H2018. This is the non-pivotal portion of the two-part study.
Although there are typical progress reports at set intervals during studies, the GBT440 HOPE Phase III trial’s top-line data is not expected until 1H2019.
Final Observations
As reported during their 3Q earnings release, the company has ~$259 million in cash with zero debt, an amount that should be sufficient to sustain operations through to the pivotal HOPE trial data. Most analyst firms appear to agree on the addressable market for GBT440 treatment and accordingly forecast peak sales of $2 billion to $3 billion. While there aren’t an abundance of catalysts between December’s ASH presentation and potential 1H2018 readout, continued positive test results would serve to strengthen the case for the company’s therapy. Although speculative, it is also possible that GBT would be a buyout candidate with its unique and effective treatment.